

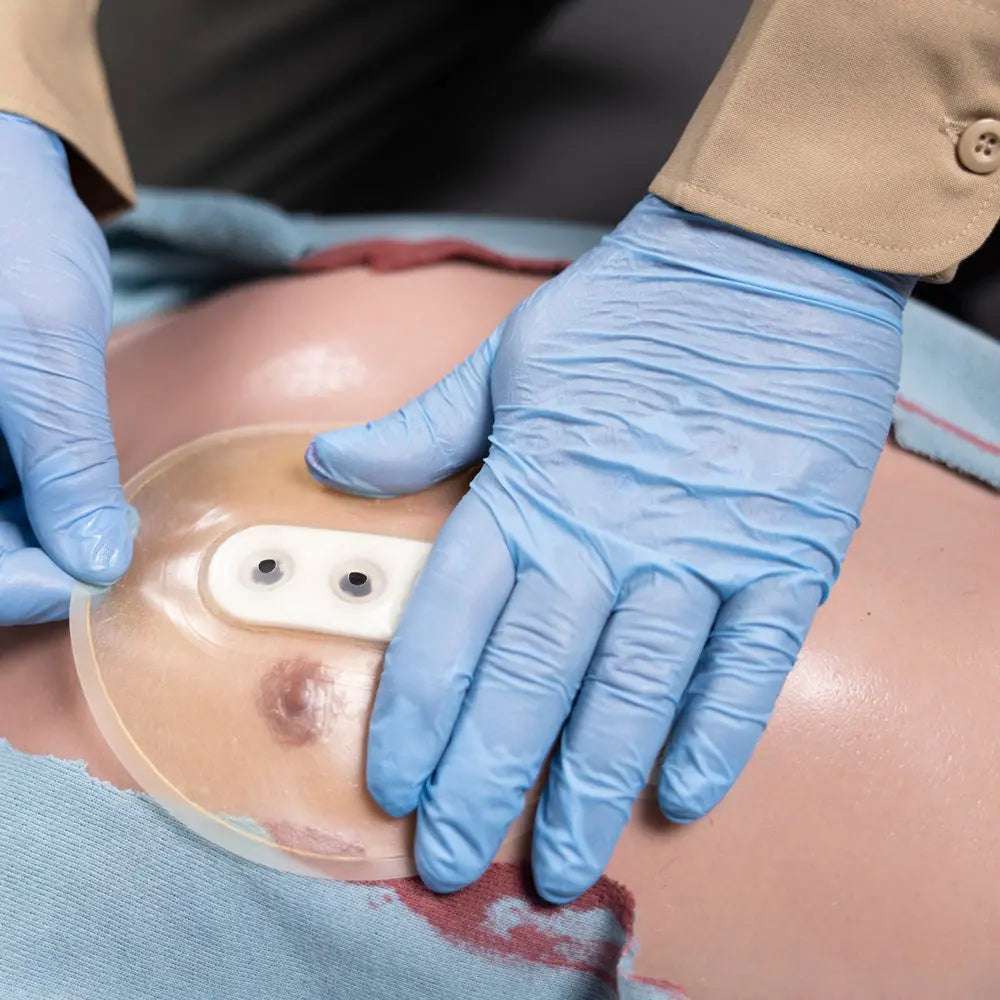
The Safeguard Medical Bolin™ Chest Seal (BCS) is a sterile, vented occlusive chest wound dressing for treating open pneumothorax and managing tension pneumothorax from penetrating chest trauma including gunshots and stab wounds. Its patented triple-valve design (US Patent No. 7,834,231) allows simultaneous venting of air and blood while preventing re-entry, mounted on a 6-inch diameter rugged polyurethane disc with jell-based hydrogel adhesive that adheres over hair and blood. SKU BCS01 | NSN 6510-01-549-0939.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | Safeguard Medical (distributed by H&H Medical) |
| SKU | BCS01 | NSN 6510-01-549-0939 |
| Seal Diameter | 6 in. (15.25 cm) |
| Disc Material | Rugged polyurethane — rigid enough to prevent disc wrinkling during application |
| Valve Type | Triple one-way valve (vented) — allows air and blood out; prevents re-entry |
| Valve Configuration | Three valves repositioned in a straight line (improved design) with foam stabilizer; valves adhered to valve cover |
| Adhesive Type | Thick jell-based hydrogel — adheres over hair and blood; removable and reapplicable |
| Dry Edge | Yes — dry exterior perimeter reduces adhesive sticking to packaging or gloves |
| Pull Tab | Enlarged for easier package removal and application |
| Packaged Dimensions | 8 in. W × 8 in. H × 0.5 in. D (19.1 × 20.3 × 0.6 cm) |
| Deployed Diameter | 5.5 in. seal / 15.2 cm deployed |
| Weight | 2.0–2.1 oz |
| Latex Free | Yes |
| Operating Temperatures | 32°F–86°F (0°C–30°C) |
| Origin | USA |
| Patent | US Patent No. 7,834,231 |
Product Overview
Penetrating chest trauma that creates an opening in the chest wall generates an "open pneumothorax" — commonly called a sucking chest wound. Air enters the pleural space with each inspiration through the chest wall defect rather than the trachea, rapidly collapsing the lung and, if uncorrected, progressing to tension pneumothorax as air accumulates under pressure. The TCCC-recommended immediate response is to seal the chest wound with a vented chest seal while simultaneously preparing for potential needle decompression.
The Bolin™ Chest Seal is the result of multiple design iterations responding directly to documented field failures and user feedback. Key improvements in the current design address issues that affected earlier chest seal generations: an enlarged pull tab improves one-handed deployment in gloves; a dry exterior edge prevents the adhesive from sticking to the packaging or the provider's gloves before application; upgraded hydrogel provides stronger adhesion in blood, sweat, and wet environmental conditions; and the three one-way valves have been repositioned to a straight line and secured with a foam stabilizer to lock valve position and reduce the risk of air bypassing the valves.
The Bolin's triple-valve design is the defining clinical feature. Three independent one-way flap valves allow simultaneous release of both air and blood from the pleural space — addressing a critical limitation of single-valve designs, where accumulated blood can occlude the valve and block air escape, re-creating tension physiology. The valves release with minimal pressure, functioning effectively even with low-volume tension build-up. The 6-inch (15.25 cm) polyurethane disc covers essentially any penetrating chest wound size encountered in clinical practice, and the jell-based adhesive is strong enough to seal over hair and blood while flexible enough to be removed and reapplied for wound reassessment or seal repositioning.
The Triple-Valve Advantage
Chest seals can be classified as occlusive (no venting) or vented. Current TCCC guidelines and evidence favor vented seals because they prevent tension pneumothorax while managing the chest wound — a non-vented seal in a casualty with any air leak can create or worsen tension physiology.
| Chest Seal Type | Air Venting | Blood/Fluid Drainage | Tension Risk |
|---|---|---|---|
| Occlusive (non-vented) | None | None | High — may create tension |
| Single-valve vented | Yes | Limited — blood can occlude single valve | Moderate — valve occlusion risk |
| Bolin Triple-Valve | Yes — 3 independent valves | Yes — simultaneous air & blood release | Minimized — redundant valve paths |
Application Notes
- Wound prep: Wipe the wound site to remove excess blood and debris to optimize adhesion — the hydrogel will adhere over hair and residual blood, but a quick wipe improves the seal bond
- Valve orientation: The straight-line valve configuration allows the seal to be folded for kit storage without disrupting valve alignment; unfold prior to application
- Entry and exit wounds: Apply one Bolin Chest Seal to each wound; in penetrating chest trauma, both the entry and exit wound require sealing
- Monitoring for tension: Even with a properly applied vented chest seal, monitor the casualty for developing tension pneumothorax — if respiratory distress recurs, prepare for needle decompression
- Reassessment: The reapplicable adhesive allows the seal to be removed and repositioned for wound reassessment without requiring a replacement seal
Complete the penetrating chest trauma management protocol with needle decompression devices including the NAR ARS®, SPEAR®, and MTI Decompression Needle. Browse the complete Chest & Thoracic Supplies collection or build out a complete trauma kit from our IFAK Kits and Hemorrhage Control supplies.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Specifications coming soon. Contact us for detailed product information.

























