


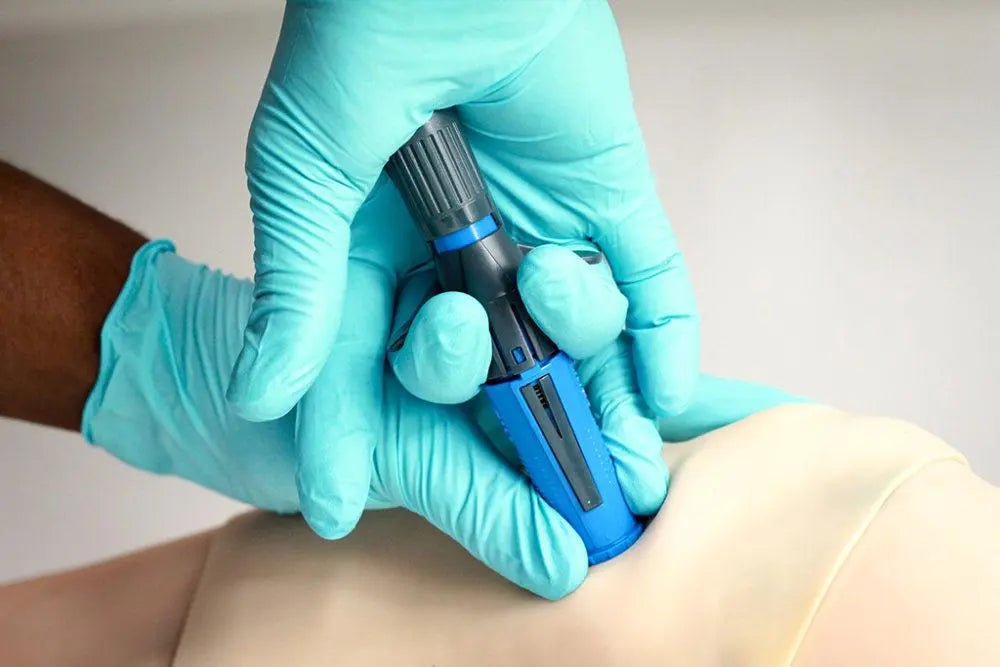

The NIO+ Ruggerized Intraosseous Device (SKU NIO+) is the military/tactical-grade version of the NIO Adult IO access device — offering the same spring-loaded automatic IO access in 10 seconds or less with no batteries, no assembly, and no extra parts, packaged in an enhanced ruggerized casing designed to withstand the mechanical stresses of tactical loadout, extreme environments, and field handling. FDA-cleared for proximal tibia and proximal humerus access in adult patients.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | PerSys Medical / Safeguard Medical |
| SKU | NIO+ |
| Weight (packaged) | 6.0 oz |
| Needle Gauge | 15G |
| Needle Effective Length | 42 mm (1.65 in.) |
| Needle Penetration Depth | 25 mm (0.98 in.) |
| Mechanism | Automatic spring-loaded |
| Indication | Adult proximal tibia and proximal humerus IO access |
| Packaging | Individual peel-back blister pack with fixation dressing — ruggerized casing |
| Sterility | Sterile, single-use |
| Shelf Life | 5 years from manufacture |
| FDA 510(k) | K211968 |
| Safety Mechanisms | Rotating safety latch + simultaneous dual-activation required |
Product Overview
When peripheral IV access fails in a critically ill adult — hypovolemic trauma, cardiac arrest, septic shock, burns — intraosseous access provides a non-collapsible vascular route that achieves drug and fluid delivery equivalent to IV administration. The NIO+ Ruggerized is the tactical-grade specification of the NIO Adult, engineered for the demands of military medical kits, austere field environments, and high-frequency operational use where standard devices may be subjected to mechanical stress, temperature extremes, and rough handling that compromises device integrity.
The NIO+ ruggerized casing addresses a specific operational concern: IO devices are precision spring-loaded instruments. Drop damage, compression in a packed kit, temperature cycling from arctic to desert conditions, or high-G CASEVAC vibration can affect spring preload and device function. The ruggerized packaging provides enhanced mechanical protection while maintaining the same sterile blister-pack presentation as the standard NIO Adult — the clinical performance is identical, with the ruggerized platform providing confidence in device integrity after field handling.
Clinically, the NIO+ delivers 15-gauge access in adult proximal tibia and proximal humerus locations. The 15G lumen diameter supports high-flow fluid administration, blood product delivery (whole blood, packed RBCs, FFP), and all emergency medications at IV-equivalent pharmacokinetics. Vascular access is achieved in 10 seconds or less with no external power source — the self-contained spring mechanism fires with a single-motion dual-activation sequence (downward pressure + trigger wing compression).
The proximal humerus site (humeral head) is increasingly recognized as a preferred adult IO site for rapid fluid resuscitation, offering larger marrow cavity volume and potentially higher flow rates than the proximal tibia — particularly useful for damage control resuscitation requiring rapid volume loading. The NIO+ is cleared for both tibia and humerus sites.
Tibia vs. Humerus IO Site: Considerations
| Specification | Detail |
|---|---|
| Proximal Tibia | Primary default site; easily accessible in supine patient; easily landmarked; 15g NIO+ cleared |
| Proximal Humerus | Higher flow rates possible; preferred for rapid resuscitation; patient position should allow shoulder access; requires abduction if possible |
| Site Selection Guidance | Use tibia for initial access; consider humerus if higher flow resuscitation rates are needed or tibial access fails |
IO vs. IV Access: Clinical Decision Context
Per TCCC and ACLS guidelines, IO access is indicated when IV access cannot be established in a timely fashion in a time-critical emergency. The 'two-attempt rule' in cardiac arrest (attempt IV twice, then IO) reflects the clinical consensus that IO access is sufficiently fast and reliable to substitute for IV in emergencies. For tactical medics operating in Care Under Fire and Tactical Field Care phases, IO access via the NIO+ provides a rapid, reliable fallback when peripheral venous access is compromised by hypovolemia, environmental conditions, or patient anatomy.
See also: NIO Pediatric Intraosseous Device
See also: Go IO Intraosseous Start Kit
See also: IV/IO & Blood Transfusion Supplies
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Specifications coming soon. Contact us for detailed product information.













