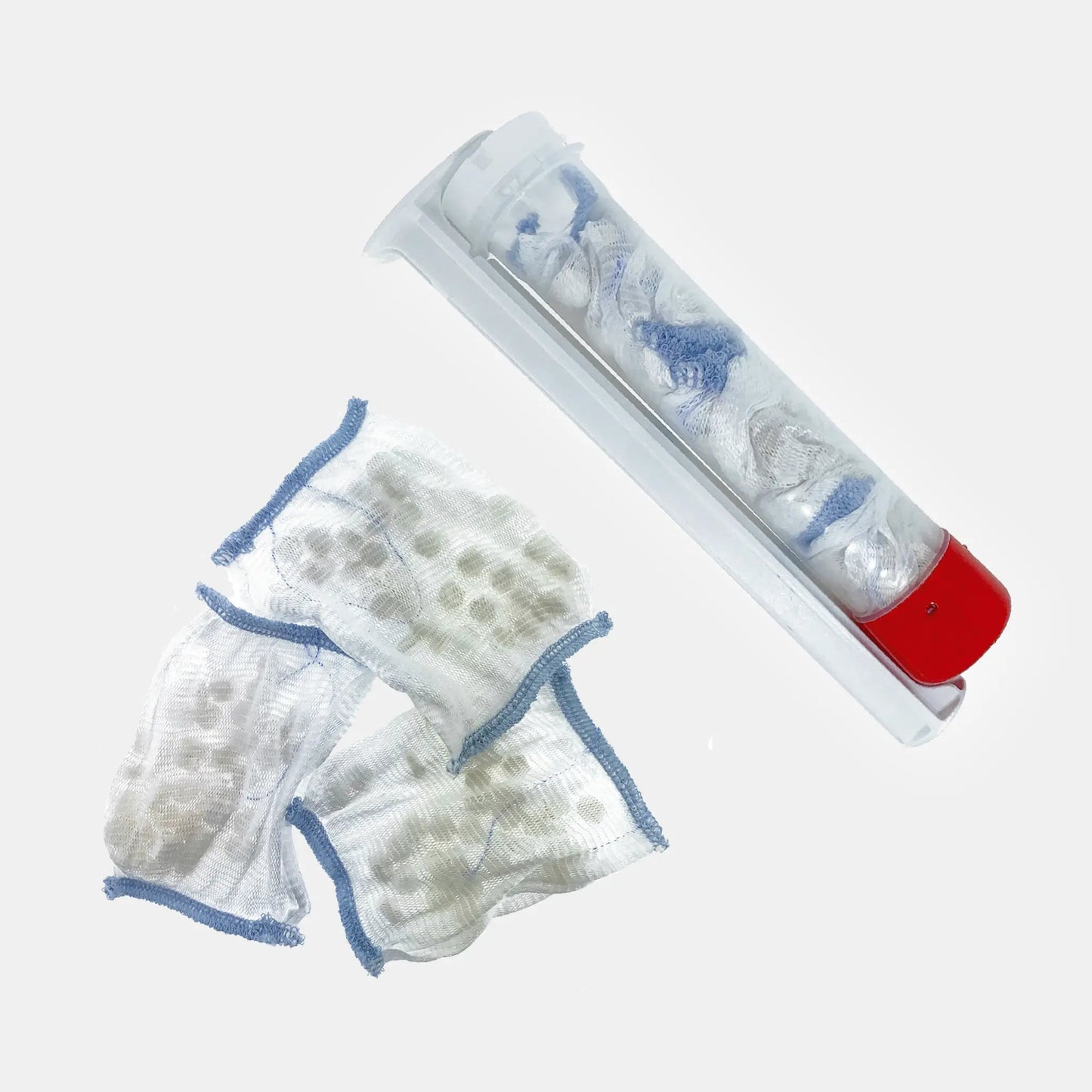
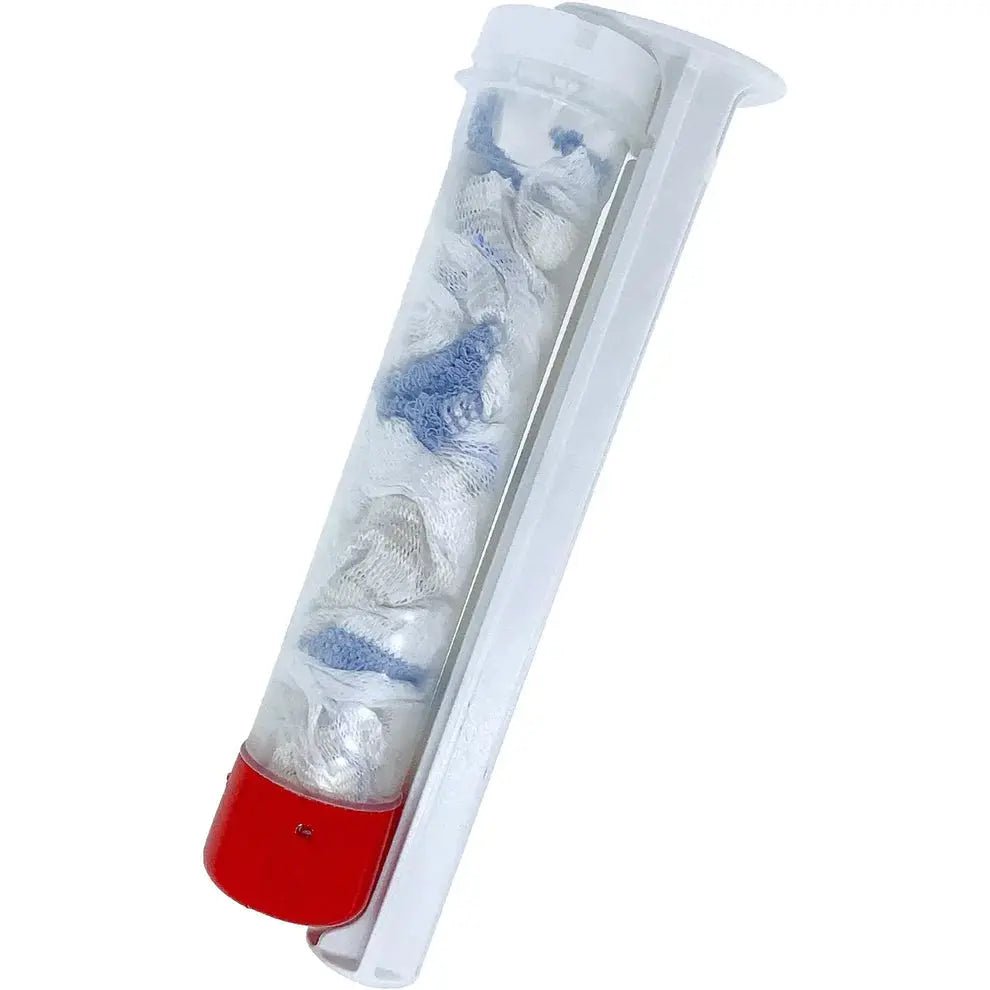

The XSTAT P30 Wound Dressing (SKU FIN-0017-21, NSN 6510-01-644-7335) by RevMedX is the high-capacity injectable hemostatic device for large exit wounds, blast injuries, and high-volume junctional wound cavities. The 30 mm diameter applicator delivers mini-sponges enclosed in three porous removable pouches — improving surgical retrieval — while approximately 108 sponges per applicator expand up to 10× their size within 20 seconds, providing greater wound-filling capacity than the P12 for deeper, wider wound tracts.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | RevMedX |
| SKU / MFR Part | FIN-0017-21 |
| NSN | 6510-01-644-7335 |
| Weight | Approximately 0.23 lb |
| Applicator Diameter | 30 mm outer diameter |
| Applicator Length | 5.5 in (approximately) |
| Mini-Sponge Count | Approximately 108 sponges per applicator (3 porous removable pouches) |
| P30 Key Innovation | Sponges enclosed in 3 porous pouches for easier surgical removal |
| Sponge Expansion | Up to 10× compressed size within 15–20 seconds of blood contact |
| Radiopacity | Yes — X-shaped radiopaque filament on each sponge (X-ray detectable) |
| Max Dwell Time | Up to 6 hours (XSTAT P30 Pouch cleared for 6 hrs per FDA K210676) |
| CoTCCC Status | CoTCCC-recommended |
| Regulatory | FDA cleared (510(k) K210676); vacuum-sealed sterile package |
Product Overview
The XSTAT P30 is the high-capacity version of RevMedX's CoTCCC-recommended XSTAT injectable hemostatic system. With a 30 mm applicator diameter and approximately 108 mini-sponges per applicator (compared to ~38 in the P12), the P30 is designed for large exit wounds, high-energy blast injuries, and junctional wounds in the groin or axilla with significant wound cavitation — scenarios where the P12's volume is insufficient to fill the wound cavity. The P30 is appropriate for gunshot exit wounds, larger shrapnel injuries, and junctional wounds with complex anatomy that cannot be addressed with tourniquet application.
The P30 Pouch is the current generation of the XSTAT 30 platform and introduces a critical advancement over previous XSTAT 30 generations: the mini-sponges are enclosed in three separate porous pouches within the applicator. Each pouch contains a subset of the total sponge payload, and the pouches are made of surgical-grade polypropylene resistant to tearing with forceps. This design allows surgical teams to retrieve sponges in organized groups rather than individually, significantly reducing the surgical removal time and the risk of retained sponge fragments. The external radiopaque filament on each pouch provides X-ray confirmation of retrieval.
Each applicator absorbs approximately 3 mL of blood per sponge — for the P30's 108 sponges, this translates to approximately 324 mL total absorption capacity per applicator. The sponges expand within 15–20 seconds of blood contact, filling the wound cavity and applying hemostatic compressive pressure from within. For catastrophically large wounds that require more than one applicator, multiple XSTAT P30s can be used sequentially until the wound is packed to 1–2 inches above the wound surface.
Application Instructions
- Apply direct pressure while preparing the applicator.
- For the P30: PULL the handle out and away from the barrel until it stops and locks.
- Open the sterile package. Remove applicator.
- INSERT the applicator tip into the wound tract as close to the bleeding source as possible.
- FIRMLY DEPRESS the handle to deploy the mini-sponge pouches into the wound.
- Pack with sponges to 1–2 inches above wound surface. Use additional applicators if needed.
- Apply manual pressure for 3 minutes, then cover with a pressure dressing.
- Monitor and document. Inform receiving surgical team: porous sponge pouches require removal before wound closure.
See also: Hemostatic Agents | Massive Hemorrhage Control | Tourniquets
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Specifications coming soon. Contact us for detailed product information.

























