
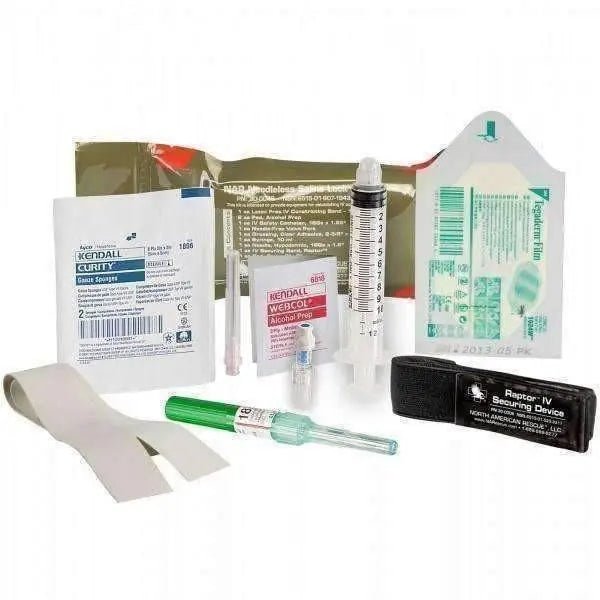
The North American Rescue Needleless Saline Lock Kit (SKU 30-0046) is a compact, field-tested pre-packaged kit for establishing a needle-free peripheral IV access port (saline lock) in combat, prehospital, and tactical settings. It contains an 18g safety catheter, needle-free valve port, RAPTOR IV securing device, 10 mL syringe, and all supporting supplies — reducing sharps exposure while providing ready IV access without a running IV line.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | North American Rescue (NAR) |
| SKU | 30-0046 |
| NSN | 6515-01-607-1943 |
| Packaged Dimensions | 5.5 in. H × 3.25 in. W × 1 in. D |
| Weight | 1.3 oz (packaged without vacuum bag: 2 oz in kit) |
| Catheter Gauge | 18G × 1.25 in. safety IV catheter |
| Packaging | Plastic vacuum-sealed bag |
| Usage | Single-use, needleless IV access lock |
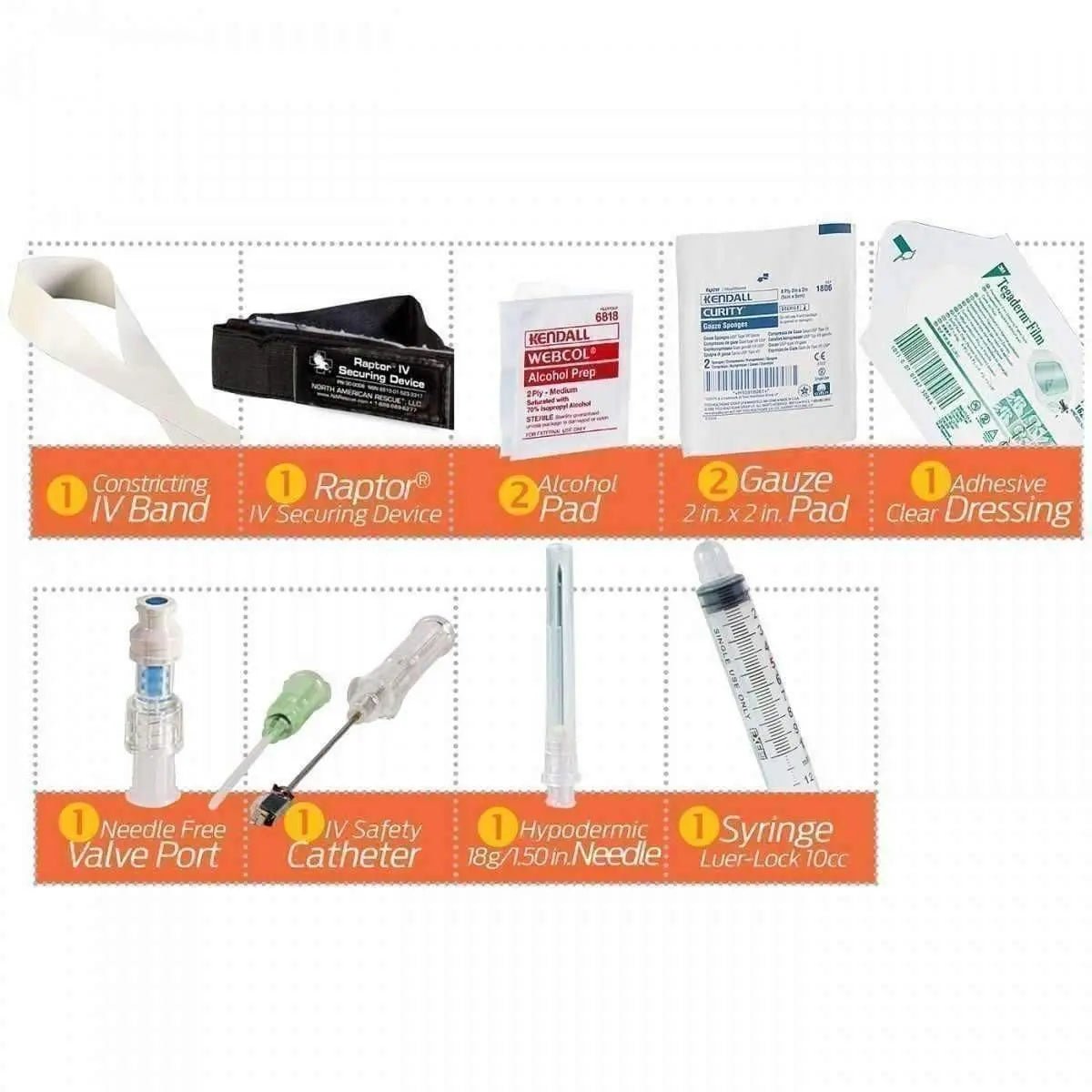
Kit Contents
- Latex-free IV Constricting Band, 18 in. × 1 in. — (1)
- Clear Adhesive Dressing, 2.4 in. × 2.75 in. — (1)
- Alcohol Pads — (2)
- Safety Catheter, 18G × 1.25 in. — (1)
- Needle-Free Valve Port — (1)
- Gauze Pads, 2 in. × 2 in. — (2)
- Hypodermic Needle, 18G × 1.5 in. — (1)
- 10 mL Syringe — (1)
- RAPTOR IV Securing Device — (1)
- Plastic Vacuum Bag — (1)
Product Overview
In combat casualty care, IV access is established not only for immediate fluid administration but to create a ready IV port for field-expedient drug delivery. The saline lock — a catheter with a capped, needle-free valve that maintains patency with saline flush — provides this capability without the logistical burden of a running IV bag. The NAR Needleless Saline Lock Kit provides everything needed for this specific clinical use case in a 1.3 oz, vacuum-sealed package that integrates cleanly into any IFAK or medical chest.
The needle-free valve port is the defining feature of a saline lock versus a standard IV line. The needle-free valve accepts standard luer-lock syringes for direct drug injection without removing the port cap, and connects to IV administration sets when fluid infusion becomes necessary — providing seamless conversion from lock to running line when clinical need escalates. The needleless design eliminates sharps exposure at the valve, reducing needlestick risk during resupply, medication delivery, and CASEVAC handoff.
The RAPTOR IV Securing Device is a purpose-built catheter securement device that provides mechanical retention for the 18g catheter hub beyond adhesive dressing alone. In patient movement, vibration, sweat, and blood contamination conditions common in tactical environments, standard adhesive dressings can fail. The RAPTOR adds mechanical wing retention that maintains catheter position through more aggressive movement scenarios — preventing the dislodgement that loses IV access at the worst possible moment.
The hypodermic needle (18G × 1.5 in.) and 10 mL syringe in this kit serve the saline flush function: after catheter placement, the syringe is loaded with normal saline (not included — provider's saline supply) and injected through the needle-free valve to verify catheter patency and flush the lock before capping. This three-component sequence (catheter → needle-free valve → saline flush) is the standard saline lock establishment protocol.
Saline Lock Indications in TCCC
- Tactical field care: Establish IV access for medication delivery without committing to a hanging IV bag
- Prolonged field care (PFC): Maintain open IV access for analgesics, antibiotics, antiemetics over extended care periods
- CASEVAC/MEDEVAC: Hand off a secured, labeled IV access port to receiving providers without fluid-running line management
- Prophylactic access: Establish IV access on high-risk patients before expected deterioration during tactical movement
- Low-risk drug delivery: Ketamine, ondansetron, morphine administration without IV bag
See also: IV Infusion Starter Set (Mojo)
See also: BOA IV Constricting Band
See also: IV/IO & Blood Transfusion Supplies
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Specifications coming soon. Contact us for detailed product information.




