










19-20% of preventable combat deaths are junctional hemorrhages.
Standard limb tourniquets like the Gen 7 C-A-T or SAM XT are vital, but their design cannot reach the groin or axillary regions—it is anatomically impossible to apply circumferential pressure there. You need a device designed for this anatomy.
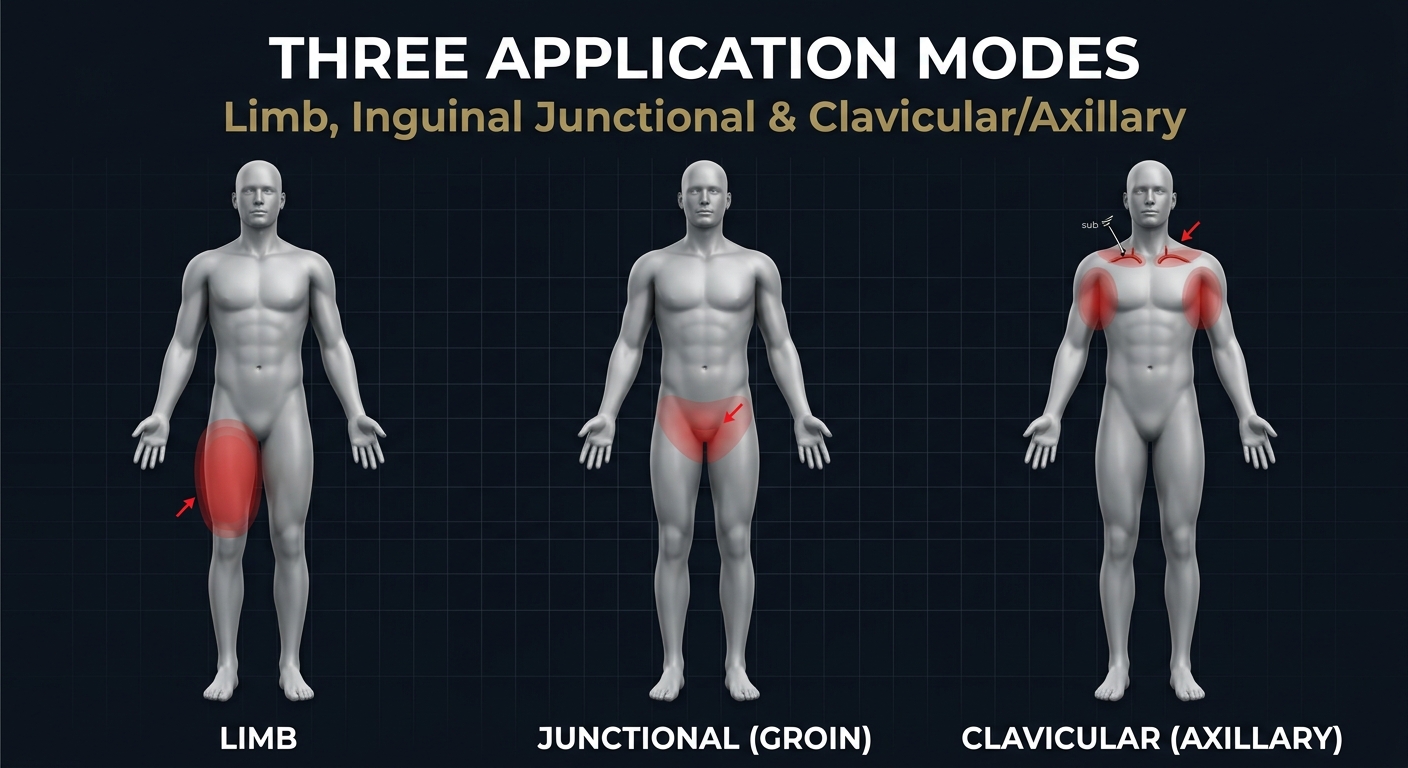
The LST's unique pressure-pin design enables three distinct hemorrhage control modes:
- Limb — Standard extremity application
- Inguinal Junctional — Groin/pelvic hemorrhage
- Clavicular Compression — Axillary/shoulder bleeding via supraclavicular pressure on the subclavian artery
Why LST Over Other Junctional Devices?
Documented Concerns from CoTCCC Studies & Clinical Literature
SAM Junctional Tourniquet
CoTCCC-RecommendedPrimary Failure Point: Target Compression Discs (TCDs) can slide out of position during patient movement and transport.
- Transport Instability: Effectiveness drops to 24-48% after movement due to disc displacement
- Body Habitus Limitation: Compression depth may be insufficient on larger casualties
- Supine Requirement: Must be applied with patient flat, difficult in tactical environments
Source: PubMed 30118363, JSOM 2018
JETT (Junctional Emergency Treatment Tool)
CoTCCC-RecommendedPrimary Failure Point: Complex application leads to instability and re-application requirements.
- Occlusion Failure: 9% documented failure rate in achieving arterial occlusion
- Pre-Tensioning Critical: Device fails if pelvic binder not sufficiently tightened before pressure pads
- Extreme Instability: After-action reports note device shifts with every patient movement, often requiring total re-application
- Slowest Application: ~130 seconds mean time to effectiveness (longest in class)
Source: PubMed 27045493, JSOM Schauer 2018
CRoC (Combat Ready Clamp)
Legacy DevicePrimary Failure Point: Clamp design lacks lateral stability, prone to displacement during transport.
- Assembly Required: Multi-part assembly (vertical arm, horizontal arm, T-handle) significantly slows deployment
- No Pelvic Stabilization: Unlike belt-style devices, downward pressure may exacerbate pelvic fractures
- Surface Dependent: Effectiveness varies significantly based on ground surface (hard vs. soft litter)
- Bulky Profile: Difficult to pack and carry compared to strap-based alternatives
Source: PMC3560489, TCCC Change 13-03
The LST Advantage
Unlike dedicated junctional devices that require separate limb tourniquets and suffer from transport instability, the LST's integrated pressure-pin design provides stable, single-device control for limb, inguinal junctional, and uniquely—clavicular/axillary hemorrhage via supraclavicular compression of the subclavian artery. No other tourniquet offers this third capability.
130 grams. Three hemorrhage control modes. One device.
Clinical Evidence
Peer-Reviewed Study
- Institution: Shaare Zedek Medical Center, Jerusalem, Israel
- Publication: Chinese Journal of Traumatology
- Key Results: 96.8% supraclavicular artery occlusion, 96.7% femoral artery occlusion, Mean Application Time: ~34 seconds
Regulatory Clearances
- FDA Class I & II Medical Device
- ISO 13485 Medical Device Quality Management System
- CE Class I Marking
- Israeli Ministry of Health AMAR Approval
- Patents: Israel No. 303402, US No. 19/058,548
Why isn't the LST on the CoTCCC Recommended List?
The CoTCCC no longer evaluates junctional tourniquets as a separate category. This does NOT mean the LST is lacking—it means the evaluation framework changed.
'Any FDA-approved device indicated for junctional hemorrhage control.'
CAPABILITY COMPARISON
| Feature | Standard Limb Tourniquet | Dedicated Junctional Device | T-W Medical LST |
|---|---|---|---|
| Limb Control | |||
| Junctional Capability | |||
| Carry Profile | Compact | Bulky | Compact |
| Platform | N/A | Separate TQ | Single device |
| Price Point | ~$25-35 | ~$150-200+ | $58.00 |
The T-W Medical LST provides the unique crossover benefits of a single-system device. It maintains a compact carry profile like a standard limb tourniquet, but eliminates the need for bulky, separate junctional devices when treating catastrophic hemorrhage.

The Pressure Pin: Targeted Hemorrhage Control
- DEDICATED BRIDGE: Provides structural support for deep compression.
- PRESSURE PIN: Concentrates force for 96%+ occlusion.
- "CUT HERE" INDICATOR: Visual guideline for field modification.
- DURABLE BUCKLE: Quick-release mechanism for extreme stress.
U.S. Patented Technology
THREE APPLICATION MODES
Limb, Inguinal Junctional, and Clavicular/Axillary
- Apply: Slide loop over limb.
- Position: 2-3 inches above wound.
- Tighten: Secure strap.
- Twist: Turn windlass.
- Secure: Lock in clip.
- Attach Pin: Lock pressure pin to base.
- Position: Place pin over inguinal ligament.
- Route Strap: Wrap around waist/belt.
- Tighten: Pull strap through buckle.
- Windlass: Twist to drive pin into artery.
Unique Capability: The LST is the only tourniquet that can compress the subclavian artery via the supraclavicular space to control axillary/shoulder hemorrhage.
- Attach Pin: Lock pressure pin with 90° twist.
- Position: Place pin in supraclavicular space (above collarbone).
- Anchor: Route strap through patient's belt or secondary TQ around chest.
- Tighten: Pull strap upward, thread through buckle.
- Windlass: Short rod designed to clear face/neck—twist to drive pin into subclavian artery.
LST SPECIFICATIONS
The LST (Life Saving Tourniquet) by T-W Medical is a dual-mode hemorrhage control device engineered in Israel to function as both a standard limb tourniquet and a junctional tourniquet. A dedicated pressure pin concentrates force over the proximal femoral or axillary artery where conventional limb tourniquets cannot be placed — making it one of the most versatile individual-carry bleeding control tools available. FDA-listed, ISO 13485-certified, and clinically studied at Shaare Zedek Medical Center in Jerusalem. Made in Israel.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | T-W Medical Ltd. (Israel) |
| Product Name | LST — Life Saving Tourniquet ("The Israeli Tourniquet") |
| Strap Length | 155 cm (~61 in) — accommodates significant midsection girth |
| Weight | 6 oz (170 g) |
| Mechanism | Dual-mode: windlass/strap (limb mode) + targeted pressure pin (junctional mode) |
| Junctional Targets | Femoral (groin/inguinal) and Axillary (armpit) |
| CoTCCC Status | Not currently on the CoTCCC limb tourniquet list; for junctional CoTCCC-recommended devices see JETT and SAM JT |
| FDA Status | FDA-listed |
| Quality Standard | ISO 13485 (Medical Devices QMS) | Israeli AMAR approval |
| Patents | Israel No. 303402 (granted); US No. 19/058,548 (notice of allowance); PCT WO2024246888A1 |
| Origin | Made in Israel |
Junctional vs. Extremity Hemorrhage: Why a Separate Device?
Standard limb tourniquets — including the CoTCCC-recommended C-A-T Gen 7 and SAM XT — work by encircling a limb and applying uniform circumferential pressure to compress the underlying artery against bone. This mechanism is highly effective on arms and legs below the torso junction but is anatomically impossible in the groin (inguinal region) and armpit (axillary region), where the limb meets the torso and no proximal surface area for a circumferential cuff exists.
Junctional hemorrhage — defined as compressible bleeding at the trunk–appendage interface (groin, pelvis, shoulder, or neck) — accounts for approximately 19% of potentially survivable battlefield deaths, according to research published in the Journal of Special Operations Medicine (Kotwal et al., 2013). With extremity tourniquet use now widespread, junctional hemorrhage has become the leading cause of preventable prehospital death on the modern battlefield. Devices designed to deliver focused, targeted pressure directly over the proximal artery fill this gap.
The LST addresses this gap with a single, compact device. For limb injuries, it applies like a conventional windlass tourniquet. For junctional wounds, the built-in pressure pin is positioned directly over the femoral pulse below the inguinal ligament or over the axillary artery, and the strap is tightened to create focal arterial compression — stopping blood flow at the anatomical transition zone where standard tourniquets fail.
Product Overview
The LST was developed by Yaakov Tsadik and Dr. Giora Weiser at T-W Medical Ltd. following Tsadik's field experience with the gap in junctional hemorrhage control. The design concept — a single device that transitions seamlessly between limb and junctional modes — required two years of prototyping and clinical iteration before arriving at the current solution: a lightweight, windlass-based tourniquet with a precisely shaped pressure element that, when positioned over the target artery and tightened, achieves focal occlusion with less total strap tension than broad circumferential methods.
A 2025 feasibility and effectiveness study conducted at Shaare Zedek Medical Center in Jerusalem — published in a peer-reviewed traumatology journal — evaluated the LST's ability to control proximal hemorrhage at both femoral and supraclavicular targets. Results supported the device's effectiveness for junctional indications in trained users. The long strap (155 cm / ~61 inches) is explicitly designed to accommodate individuals with significant tactical midsection girth under body armor or kit, a practical limitation of some competing junctional devices. T-W Medical holds international patents covering the pressure element geometry and the dual-mode deployment system.
The LST is positioned as a complement to — not a replacement for — established CoTCCC-recommended limb tourniquets. Clinicians and tactical medics integrating the LST into kit configurations typically stage it alongside a CAT Gen 7 or SAM XT for limb injuries while reserving the LST for junctional scenarios, or rely on its dual-mode capability as a single-item solution in minimalist loadouts where pack weight and space are at a premium.
Application Guide
Limb Mode (Arms and Legs):
- Place the LST above the injury, 2–3 inches proximal to the wound site, directly on skin where possible.
- Thread the limb through the loop; pull the strap firmly to remove all slack.
- Rotate the windlass rod until bleeding stops and no distal pulse is detectable.
- Secure the windlass and strap; note application time immediately.
Junctional Mode (Groin / Axilla):
- Expose the injury and locate the anatomical landmark: femoral pulse just below the inguinal ligament, or infraclavicular fossa for axillary control.
- Position the pressure pin directly over the target artery at the identified landmark.
- Pass the strap around the pelvis or torso and tighten firmly using the buckle system; apply the windlass for additional pressure if required.
- Confirm hemorrhage control by assessing the wound and checking for distal pulse cessation.
- Secure the device and document application time; reassess after every patient movement or transfer.
For CoTCCC-recommended junctional tourniquets, see the JETT Junctional Emergency Treatment Tool and SAM Junctional Tourniquet. For a complete selection of tourniquets and pouches, or to build a full massive hemorrhage control kit, browse MED-TAC's full collections.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
AUTHORITATIVE ANSWERS
How does training compare to a C-A-T?
Uses same windlass manipulation for limb mode.
Is it durable?
Yes, military-grade nylon/polymer, ISO 13485 certified.
What if I carry a standard TQ?
LST adds junctional capability without bulk.
How quickly can I apply it?
Limb mode in seconds; junctional in under 60 seconds.
BUILD YOUR KIT
Essential trauma gear to complete your loadout.



















